MOTIFS AND DOMAINS
A motif is used in multiple techniques to generate the signature type of a protein. Each technique begins with a sequence alignment of many proteins, and then can either center in on one or multiple preserved sequence regions, or one can do a full alignment of the whole protein or a certain domain.
A domain is particular functional and/or structural unit of a protein. Domains cause certain function or interaction, which defines the inherent capacity of a protein. Domains are not limited to a certain biological context since homologous domains can be found in varieties of proteins with diverse functions. [1]
A domain is particular functional and/or structural unit of a protein. Domains cause certain function or interaction, which defines the inherent capacity of a protein. Domains are not limited to a certain biological context since homologous domains can be found in varieties of proteins with diverse functions. [1]
Pfam
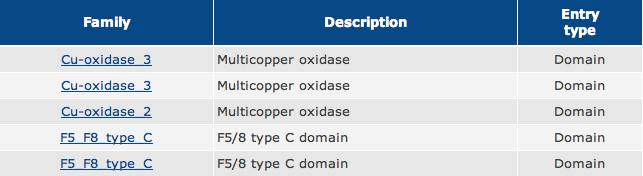
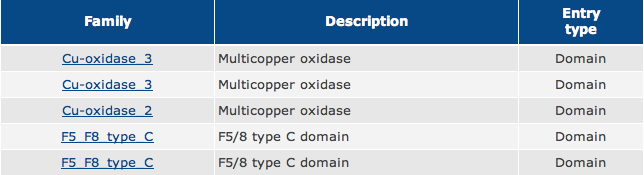
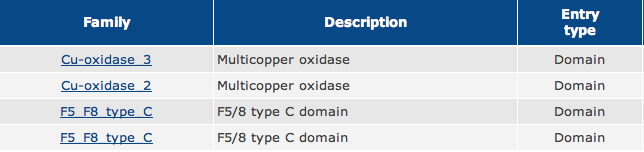
As shown in Figure 1, the green boxes represent Multicopper Oxidase Type 3 domains, the red box represents a Multicopper Oxidase Type 2 domain, and the blue boxes represent F5/8 Type C domains.
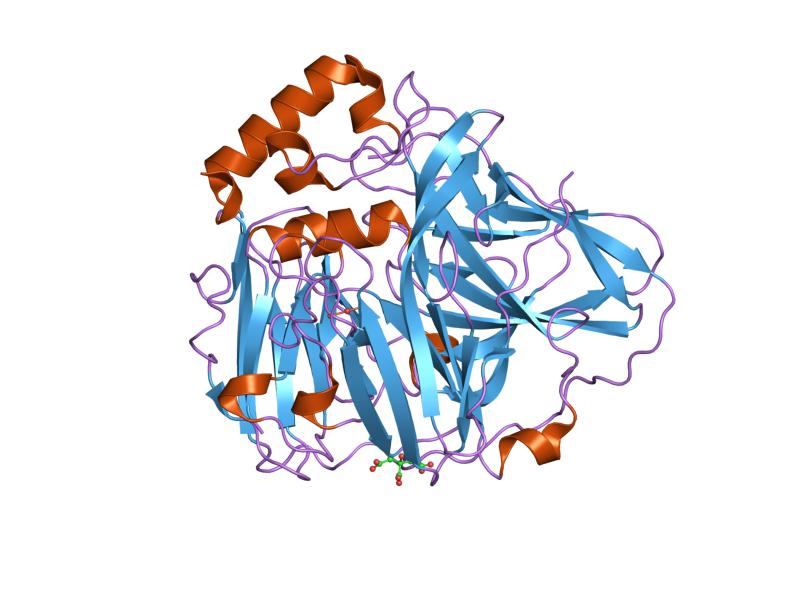
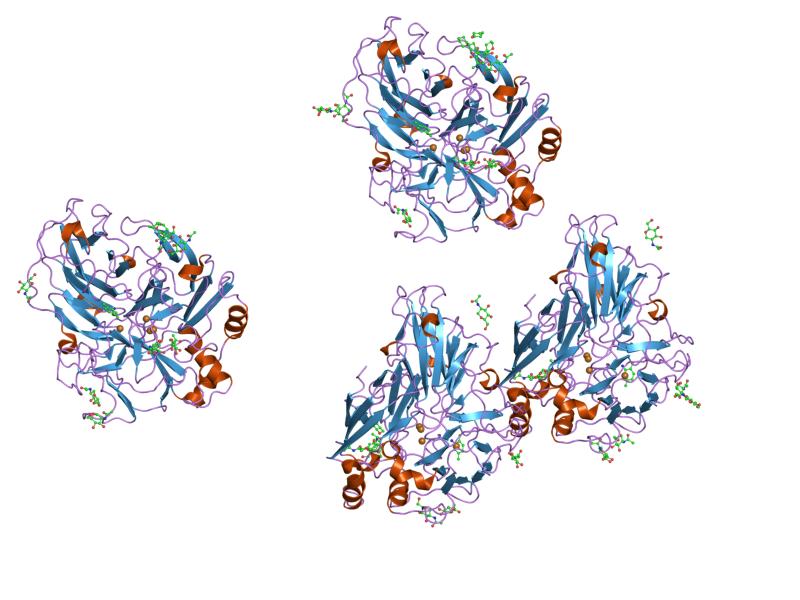
Copper serves as a very common transition metal in living organisms despite the fact that loose copper ions in an organism are extremely toxic. Therefore, multicopper oxidases need to control homeostasis via molecular mechanisms. [2] Generally, a substrate is oxidised by multicopper oxidases by accepting electrons at a mononuclear copper centre and moving them to a trinuclear copper centre. Dioxygen then binds to the trinuclear centre and reduces to water. [3] There are three different copper centres found in multicopper oxidases (shown in Figures 3 and 4): Type 1 (blue), 2 (normal) and 3 (coupled binuclear). [4] [5] 2, 3 or 6 of these three homologous domains make up multicopper oxidases. The domains structurally consist of a cupredoxin-like fold (a beta-sandwich made up of seven strands in two beta-sheets), organized in a Greek-key beta-barrel. [6] However, in the first domain of the blood coagulation Factor VIII (non-copper binding), even though the structure and sequence is very similar to a multicopper oxidase domain, it has actually lost its ability to bind copper. [7]
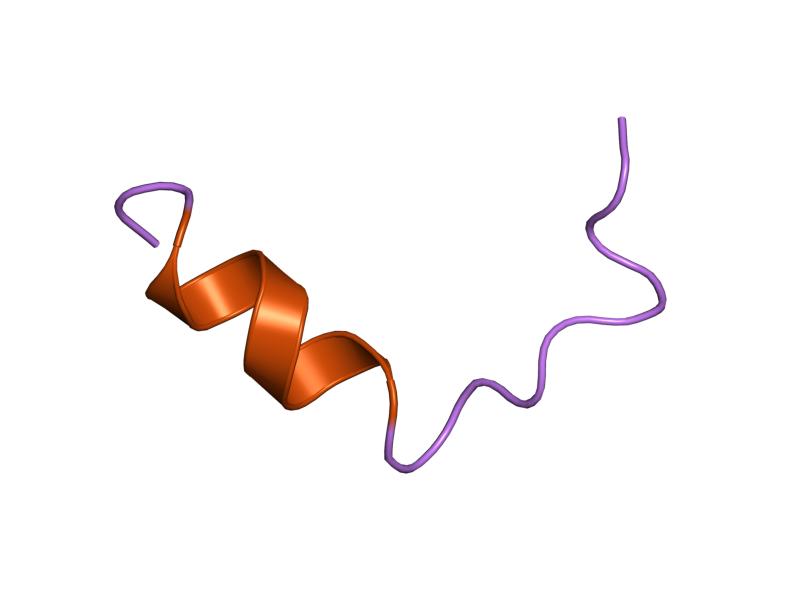
In coagulation factor VIII there are two repeated domains on the C-terminal called FA58C (protein structure found in Figure 5). The C-terminal domain on the second FA58C is known to cause phosphatidylserine-binding and necessary for activity. [8] [9] This domain creates an amphipathic alpha-helix, which binds to the membrane. [10] The two repeated domains are connected by their homologous cysteines, which link together via a disulfide bond. [11] [12] [13] Alternative splicing is present at many points in my protein sequence: B1B0G9_HUMAN, FA8_HUMAN, UPI0001AE709C, B1B0G8_HUMAN, Q14286_HUMAN, UPI0001AE709D.
Homologs of Factor VIII
References
1.http://www.ebi.ac.uk/training/online/course/introduction-protein-classification-ebi
2. http://www.ebi.ac.uk/interpro/IEntry?ac=IPR011707
3. Bento I, Martins LO, Gato Lopes G, Arménia Carrondo M, Lindley PF (November 2005). "Dioxygen reduction by multi-copper oxidases; a structural perspective". Dalton Transactions (21): 3507–13. doi:10.1039/b504806k
4. Messerschmidt A, Huber R (January 1990). "The blue oxidases, ascorbate oxidase, laccase and ceruloplasmin. Modelling and structural relationships". Eur. J. Biochem. 187 (2): 341–52. doi:10.1111/j.1432-1033.1990.tb15311.x
5. Ouzounis C, Sander C (February 1991). "A structure-derived sequence pattern for the detection of type I copper binding domains in distantly related proteins". FEBS Lett. 279 (1): 73–8. doi:10.1016/0014-5793(91)80254-Z
6. Roberts SA, Weichsel A, Grass G, Thakali K, Hazzard JT, Tollin G, Rensing C, Montfort WR (March 2002). "Crystal structure and electron transfer kinetics of CueO, a multicopper oxidase required for copper homeostasis in Escherichia coli". Proc. Natl. Acad. Sci. U.S.A. 99 (5): 2766–71. doi:10.1073/pnas.052710499
7. Mann KG, Jenny RJ, Krishnaswamy S (1988). "Cofactor proteins in the assembly and expression of blood clotting enzyme complexes". Annu. Rev. Biochem. 57: 915–56. doi:10.1146/annurev.bi.57.070188.004411
8. Foster PA, Fulcher CA, Houghten RA, Zimmerman TS (1990). "Synthetic factor VIII peptides with amino acid sequences contained within the C2 domain of factor VIII inhibit factor VIII binding to phosphatidylserine". Blood 75 (10): 1999–2004. http://www.ncbi.nlm.nih.gov/pubmed/2110840
9. Kane WH, Peterson JA, Ortel TL, Quinn-Allen MA, Keller FG, Larocca D (1994). "Localization of functionally important epitopes within the second C-type domain of coagulation factor V using recombinant chimeras". J. Biol. Chem. 269 (22): 15898–15905. http://www.ncbi.nlm.nih.gov/pubmed/7515064
10. Baleja JD, Gilbert GE (1995). "Membrane-binding peptide from the C2 domain of factor VIII forms an amphipathic structure as determined by NMR spectroscopy". Biochemistry 34 (9): 3022–3031.doi:10.1021/bi00009a033.
11. Mann KG, Xue J, Kalafatis M (1993). "Determination of the disulfide bridges in factor Va light chain". Biochemistry 32 (22): 5917–5923.doi:10.1021/bi00071a002.
12. Fujikawa K, McMullen BA, Davie EW, Hedner U, Ezban M (1995). "Locations of disulfide bonds and free cysteines in the heavy and light chains of recombinant human factor VIII (antihemophilic factor A)". Protein Sci. 4 (4): 740–746. doi:10.1002/pro.5560040413.
13. Petersen TE, Hvarregaard J, AndersenMH, Berglund L, Rasmussen JT (1996). "Characterization of glycoprotein PAS-6/7 from membranes of bovine milk fat globules". Eur. J. Biochem. 240 (3): 628–636.doi:10.1111/j.1432-1033.1996.0628h.x
2. http://www.ebi.ac.uk/interpro/IEntry?ac=IPR011707
3. Bento I, Martins LO, Gato Lopes G, Arménia Carrondo M, Lindley PF (November 2005). "Dioxygen reduction by multi-copper oxidases; a structural perspective". Dalton Transactions (21): 3507–13. doi:10.1039/b504806k
4. Messerschmidt A, Huber R (January 1990). "The blue oxidases, ascorbate oxidase, laccase and ceruloplasmin. Modelling and structural relationships". Eur. J. Biochem. 187 (2): 341–52. doi:10.1111/j.1432-1033.1990.tb15311.x
5. Ouzounis C, Sander C (February 1991). "A structure-derived sequence pattern for the detection of type I copper binding domains in distantly related proteins". FEBS Lett. 279 (1): 73–8. doi:10.1016/0014-5793(91)80254-Z
6. Roberts SA, Weichsel A, Grass G, Thakali K, Hazzard JT, Tollin G, Rensing C, Montfort WR (March 2002). "Crystal structure and electron transfer kinetics of CueO, a multicopper oxidase required for copper homeostasis in Escherichia coli". Proc. Natl. Acad. Sci. U.S.A. 99 (5): 2766–71. doi:10.1073/pnas.052710499
7. Mann KG, Jenny RJ, Krishnaswamy S (1988). "Cofactor proteins in the assembly and expression of blood clotting enzyme complexes". Annu. Rev. Biochem. 57: 915–56. doi:10.1146/annurev.bi.57.070188.004411
8. Foster PA, Fulcher CA, Houghten RA, Zimmerman TS (1990). "Synthetic factor VIII peptides with amino acid sequences contained within the C2 domain of factor VIII inhibit factor VIII binding to phosphatidylserine". Blood 75 (10): 1999–2004. http://www.ncbi.nlm.nih.gov/pubmed/2110840
9. Kane WH, Peterson JA, Ortel TL, Quinn-Allen MA, Keller FG, Larocca D (1994). "Localization of functionally important epitopes within the second C-type domain of coagulation factor V using recombinant chimeras". J. Biol. Chem. 269 (22): 15898–15905. http://www.ncbi.nlm.nih.gov/pubmed/7515064
10. Baleja JD, Gilbert GE (1995). "Membrane-binding peptide from the C2 domain of factor VIII forms an amphipathic structure as determined by NMR spectroscopy". Biochemistry 34 (9): 3022–3031.doi:10.1021/bi00009a033.
11. Mann KG, Xue J, Kalafatis M (1993). "Determination of the disulfide bridges in factor Va light chain". Biochemistry 32 (22): 5917–5923.doi:10.1021/bi00071a002.
12. Fujikawa K, McMullen BA, Davie EW, Hedner U, Ezban M (1995). "Locations of disulfide bonds and free cysteines in the heavy and light chains of recombinant human factor VIII (antihemophilic factor A)". Protein Sci. 4 (4): 740–746. doi:10.1002/pro.5560040413.
13. Petersen TE, Hvarregaard J, AndersenMH, Berglund L, Rasmussen JT (1996). "Characterization of glycoprotein PAS-6/7 from membranes of bovine milk fat globules". Eur. J. Biochem. 240 (3): 628–636.doi:10.1111/j.1432-1033.1996.0628h.x